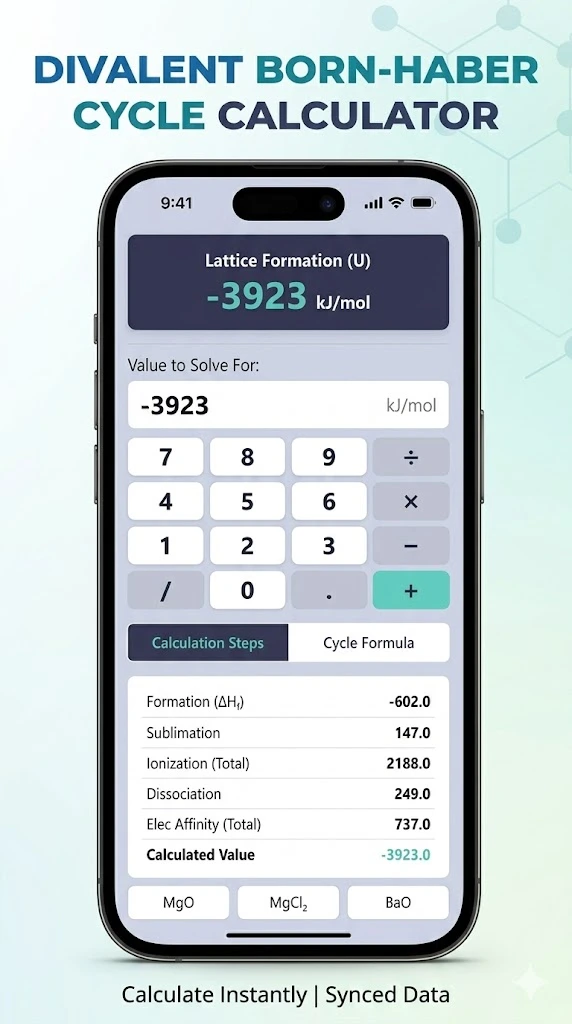
Divalent Born Haber Cycle Calculator
Stoichiometry Matters! For compounds like MgCl2, your IE, EA, and Dissociation terms must reflect the final mole count of the product (e.g., use 2x the Cl terms).
kJ/mol
Full Divalent Hess Loop
The calculator automatically rearranges this algebraic expression to isolate your selected target variable.
Calculate Lattice Energy Using Divalent Born-Haber Cycle Too
The Born-Haber Cycle is a widely used thermochemical cycle named after scientists Max Born and Fritz Haber. It primarily serves as a method to calculate the Lattice Energy of an ionic solid—a value that is notoriously difficult to measure directly through physical experiments.
While standard monovalent cycles calculate energies for simple compounds like Table Salt (NaCl), this tool handles the more complex divalent cycle. This is essential for compounds formed from +2 cations and -2 anions (like MgO) or compounds involving multiple ion moles (like MgCl2). By applying Hess’s Law, this tool lets you algebraically solve for any single missing thermodynamic variable in a full formation loop.
Lattice Energy Calculation Formula & Example
To specifically calculate Lattice Energy (U), we rearrange the general Born-Haber equation to isolate U on one side of the equation:
Example Calculation: Calcium Chloride (CaCl2)
Let’s calculate the Lattice Energy of Calcium Chloride. Note the stoichiometry: we need one Calcium ion (Ca2+) and two Chloride ions (2Cl–).
- Enthalpy of Formation (ΔHf) of CaCl2 = -796 kJ/mol
- Sublimation (ΔHsub) of Ca = +178 kJ/mol
- Ionization Energies (IE1 + IE2) of Ca = 590 + 1145 = +1735 kJ/mol
- Bond Dissociation (ΔHdiss) of Cl2 = +242 kJ/mol (Breaking 1 mole of Cl2 gas perfectly yields the 2 moles of Cl atoms we need).
- Electron Affinity (EA) of Cl = -349 kJ/mol. (Since we need 2 moles of Cl ions, total EA = 2 × -349 = -698 kJ/mol).
Applying the formula:
U = -796 – [178 + 1735 + 242 + (-698)]
U = -796 – [1457]
U = -2253 kJ/mol (The negative sign confirms that energy is released when the strong ionic crystal lattice forms).
Breakdown of the Cycle Components
| Term | Full Name | Process (Example Mg + O) | Typical Sign |
|---|---|---|---|
| ΔHf | Enthalpy of Formation | Mg(s) + 1/2 O2(g) → MgO(s) | Exothermic (-) |
| ΔHsub | Sublimation | Mg(s) → Mg(g) | Endothermic (+) |
| IE1 & IE2 | Ionization Energies (1 & 2) | Mg(g) → Mg+(g) → Mg2+(g) | Endothermic (+) |
| ΔHdiss | Bond Dissociation | 1/2 O2(g) → O(g) | Endothermic (+) |
| EA1 | Electron Affinity 1 | O(g) + e– → O–(g) | Exothermic (-) |
| EA2 | Electron Affinity 2 | O–(g) + e– → O2-(g) | Endothermic (+) |
| U | Lattice Formation Energy | Mg2+(g) + O2-(g) → MgO(s) | Exothermic (-) |
Applications of Born–Haber Cycle in Daily Life
While it seems like purely theoretical chemistry, the Born-Haber cycle and the lattice energies it calculates have profound impacts on the modern products we use every day:
- Next-Gen Battery Technology: Engineers use lattice energy calculations to design solid-state electrolytes for lithium-ion batteries. A lower lattice energy means ions can move more freely, leading to batteries that charge faster and hold more power for electric vehicles and smartphones.
- Pharmaceutical Design: The lattice energy of an active pharmaceutical ingredient (API) in a pill directly affects its solubility. Drug designers use these principles to ensure a pill breaks down in the stomach at the exact rate needed for the body to absorb it safely.
- Advanced Ceramics & Materials: Compounds with incredibly high lattice energies (like Aluminum Oxide) are highly stable and heat resistant. This helps in manufacturing heat shields for spacecraft, durable engine components, and cutting tools.
- Water Softening & Detergents: Understanding how easily calcium and magnesium ions break away from their solid forms helps chemists design better water softeners and industrial detergents that prevent scale buildup in pipes.
Common Pitfalls to Avoid (Pro Tips)
- Mind your signs: Remember that taking an electron away (Ionization) always requires energy (+), while giving an electron (1st Electron Affinity) usually releases energy (-). However, forcing a second electron onto an already negative ion (2nd Electron Affinity) requires energy (+).
- Diatomic Gases: When dealing with halogens (Cl2, F2, Br2) or Oxygen (O2), pay close attention to dissociation. If you need 2 moles of Cl atoms, you need to break 1 full mole of Cl2 bonds.
Frequently Asked Questions (FAQ)
Why is Lattice Energy so hard to measure directly?
Lattice energy is defined as the energy released when gaseous ions come together to form a solid. In the real world, it is practically impossible to isolate bare, gaseous ions in a vacuum and measure the heat they release as they crystallize. The Born-Haber cycle circumvents this by using Hess’s Law to add up measurable steps to find the unmeasurable one.
Why is the Second Electron Affinity (EA2) positive?
While adding an electron to a neutral atom (like Oxygen) releases energy (-), adding a second electron to an O– ion requires forcing a negative electron toward an already negative particle. Like repels like, so overcoming this electrostatic repulsion requires an input of energy (+).
Expertise, Authoritativeness, and Key References
This calculator relies on standard thermodynamic data types and the mathematical definitive proof of Hess’s Law. For strict verification, users are encouraged to consult trusted physical property databases and peer-reviewed literature.
- NIST Chemistry WebBook (SRD 69): The definitive US government resource for obtaining standardized Ionization Energies, Electron Affinities, and Enthalpy data.
- Chemistry LibreTexts: The Born-Haber Cycle: Authoritative open-access academic text detailing the algebraic proof of Hess’s Law applied to ionic solids.
- Original Work by Max Born and Fritz Haber (1919): Though published in German (Verhandlungen der Deutschen Physikalischen Gesellschaft), these foundational papers established the exact methodology still utilized in this calculator today.
Frequently Asked Questions (FAQs)
What is the Divalent Born-Haber Cycle?
The Divalent Born-Haber Cycle is a thermodynamic process used to calculate the lattice energy of ionic compounds where ions have a charge of +2 or -2. It breaks the formation of an ionic solid into multiple energy steps.
What is lattice energy in the Born-Haber Cycle?
Lattice energy is the energy released when gaseous ions combine to form an ionic solid. In the Born-Haber Cycle, it is calculated indirectly using Hess’s Law.
How is the Born-Haber Cycle different for divalent compounds?
In divalent compounds, additional ionization energies (second ionization energy) and electron affinities must be considered, making the calculation slightly more complex than monovalent cases.
What formula is used in the Divalent Born-Haber Cycle?
[latex]\text{Lattice Energy = ΔHf – (Sublimation Energy + Ionization Energies + Bond Dissociation Energy + Electron Affinity)}[/latex]
What values are required to calculate lattice energy?
You typically need:
Enthalpy of formation (ΔHf)
Sublimation energy
Ionization energies (first and second)
Bond dissociation energy
Electron affinity values
What is an example of a divalent ionic compound?
Common examples include:
MgO (Magnesium Oxide)
CaCl₂ (Calcium Chloride)
ZnO (Zinc Oxide)
What is the main purpose of the Born-Haber Cycle?
The main purpose of the Born-Haber Cycle is to calculate lattice energy using known thermodynamic data through Hess’s Law.